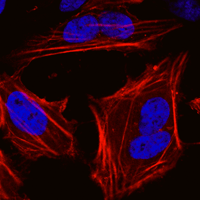
Lentiviral particles overview
We have partnered with Flash Therapeutics to provide ready-made lentiviral particles for use in research applications involving tet-inducible expression or fluorescent reporters. The exceptional quality of these premade lentiviral particles is due to a unique proprietary method for producing high-purity, high-titer particles with superior performance.
What you can expect with lentiviral particles from Flash Therapeutics:
- 100% transduction of many primary cell types (including neurons, endothelial cells, and stem cells)
- High expression in primary cells when delivered at a high multiplicity of infection (MOI)
- No cytotoxic effects
- Functional titer—not just a particle count
Unlocking the special benefits of our exclusive lentiviral particles
Unlike ready-to-use lentiviral particles from other suppliers, the particles in our catalog are produced via a multi-step purification process, which results in very high titers that increase transduction efficiency. The high purity reduces cytotoxicity, so you can successfully transduce a large number of different cell types—including primary, immortalized, adherent, and suspension cells—while retaining high cell viability.
State-of-the-art lentivirus production platform
Flash Therapeutics has developed and patented a unique process for the production of high-titer viral supernatant. This process involves generation of virus in HEK293T cells, followed by back-to-back tangential ultrafiltration with diafiltration to concentrate and purify the particles. Finally, after titration and sterility testing, high-titer, high-purity lentiviral vectors are ready for transduction of your cells.
Figure 1. Overview of the process for generating high-titer lentiviral particles. Through this proprietary, large-scale process, Flash Therapeutics is able to produce very high-titer, high-purity lentiviruses ready for transduction of many different cell types.
Enhance your research with our functional titer
To facilitate project success, we supply each batch of lentiviral particles with a functional titer of >1 x 109 transduction units per ml (TU/ml). Unlike other viral suppliers, we don’t just provide particle counts but guarantee a functional titer, ensuring only the highest quality virions with the ability to integrate into cells. Our cutting-edge methodology surpasses industry standards, resulting in up to a 100-fold increase in infectious particles compared to other vendors.

Elevated titers enhance the speed, simplicity, and effectiveness of lentiviral transductions. Increased lentiviral titers enable the efficient infection of challenging cell types even at lower Multiplicity of Infection (MOI), while preserving the initial phenotype. The exceptional purity of our lentiviral particles significantly reduces cytotoxicity, further amplifying the transduction efficiency in delicate cells.

Figure 2. Transduction efficiency of a Flash Therapeutics ZsGreen-expressing lentiviral vector in T cells. Abbreviations: RFU = relative fluorescence units.

Figure 3. High concentration and high purification of Flash Therapeutics’ lentiviral vectors ensure optimal transduction efficiency across all cell types (immortalized and primary cells). Abbreviations: iPSCs = induced pluripotent stem cells; HSCs = hematopoietic stem cells.
Products available from Takara Bio
Viral particles for Tet-inducible gene expression
We offer ready-made lentiviral particles to introduce constitutive expression of the Tet-On 3G transactivator in your cells of choice. In the presence of doxycycline (a tetracycline derivative), the Tet-On 3G transactivator binds to and activates transcription of the TRE3G promoter (PTRE3G). This allows you to precisely control transcription of any gene cloned downstream of PTRE3G. Learn more »
Viral particles for subcellular localization studies
Constitutive lentiviral reporter vectors are prepackaged lentiviral particles that deliver fluorescent proteins targeted to specific organelles or subcellular structures. These particles can be used to create reporter cell lines for the study of cytoskeletal and organelle structure and function in living cells or protein localization studies. Learn more »
Schematic maps of expression plasmids used for preparation of pre-packaged, high concentration lentivirus particles. Click on the images to view larger versions.
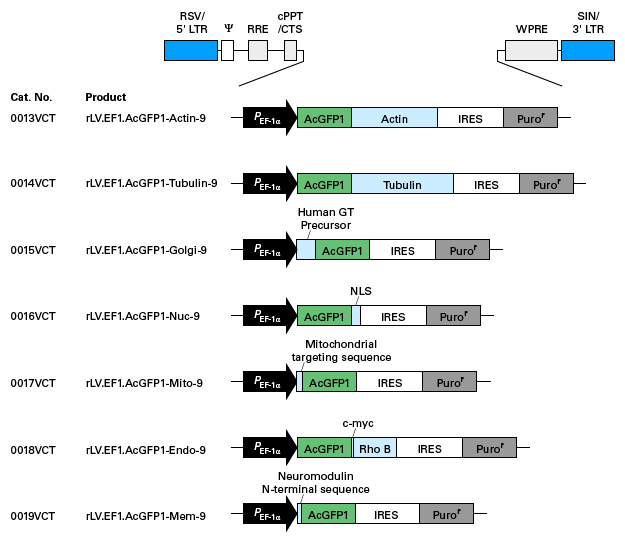
Expression plasmids for labeling cellular compartments with GFP
Plasmids for creating lentiviral particles for labeling of cellular compartments (green)
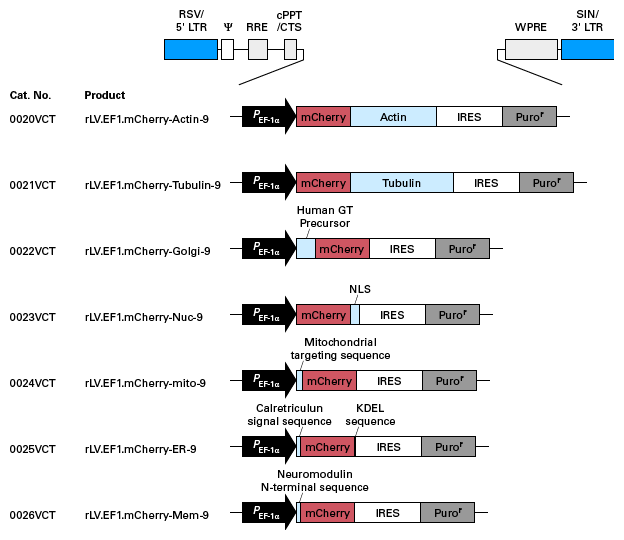
Expression plasmids for labeling cellular compartments with mCherry
Plasmids for creating lentiviral particles for labeling of cellular compartments (red)

Expression plasmids for whole-cell labeling
Plasmids for creating lentiviral particles for whole-cell labeling (multiple colors)
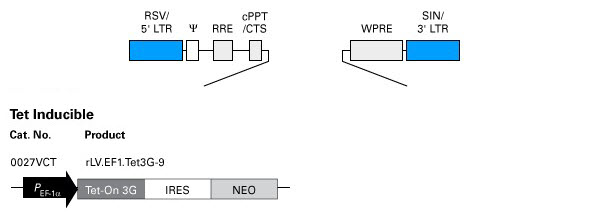
Expression plasmid for inducible expression
Plasmid for creating lentiviral particles for inducible expression
Takara Bio USA, Inc.
United States/Canada: +1.800.662.2566 • Asia Pacific: +1.650.919.7300 • Europe: +33.(0)1.3904.6880 • Japan: +81.(0)77.565.6999
FOR RESEARCH USE ONLY. NOT FOR USE IN DIAGNOSTIC PROCEDURES. © 2025 Takara Bio Inc. All Rights Reserved. All trademarks are the property of Takara Bio Inc. or its affiliate(s) in the U.S. and/or other countries or their respective owners. Certain trademarks may not be registered in all jurisdictions. Additional product, intellectual property, and restricted use information is available at takarabio.com.